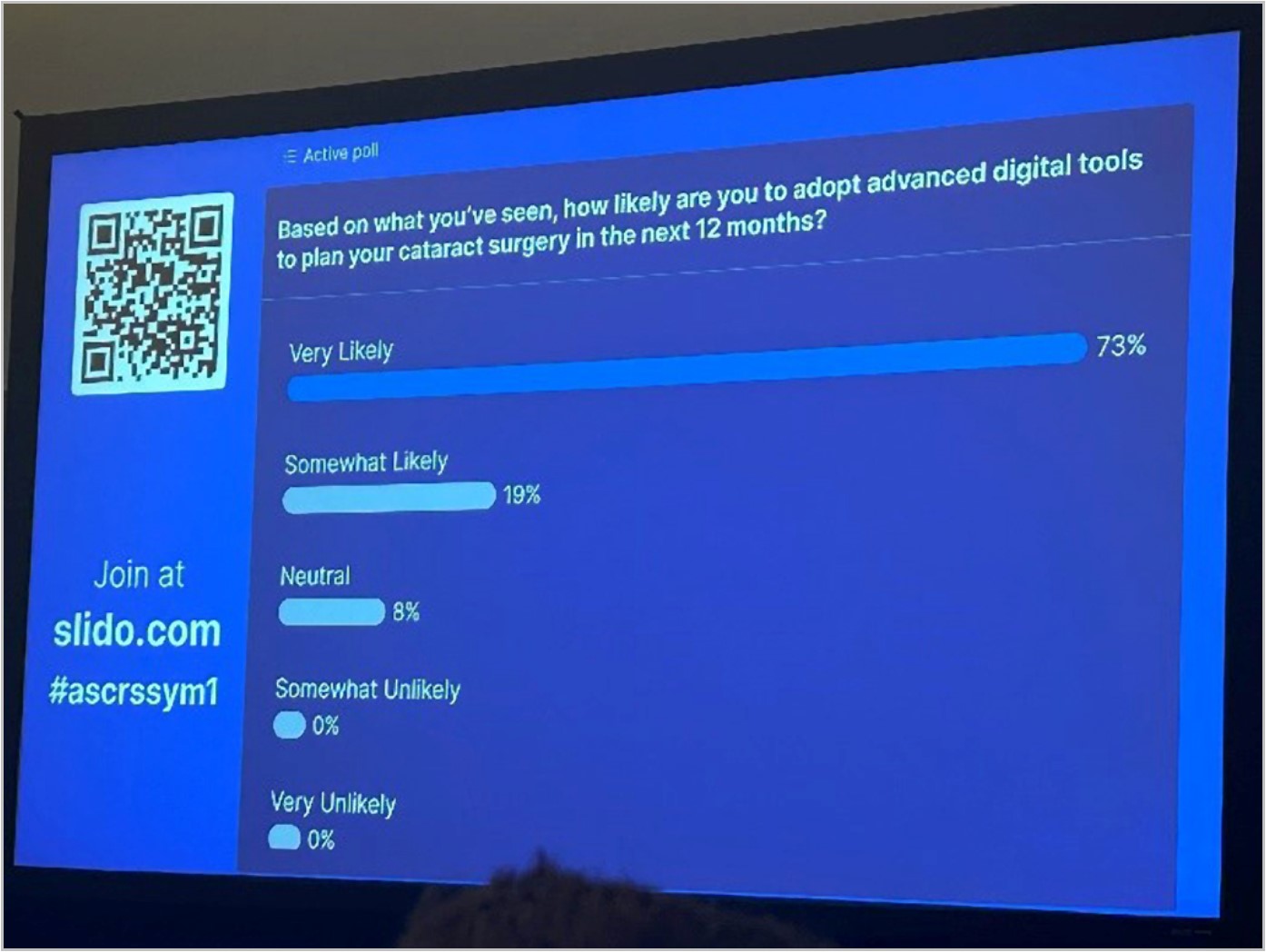
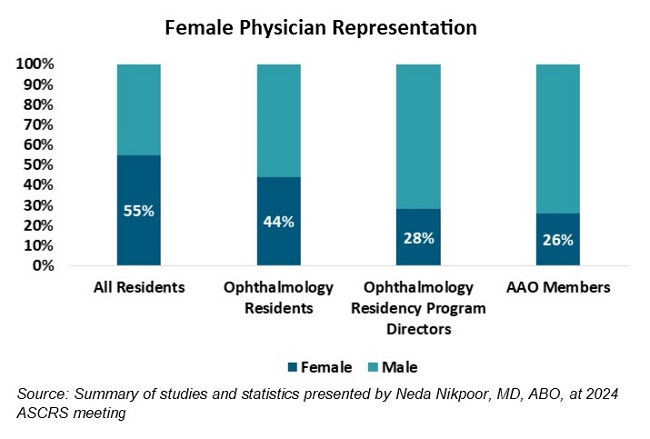
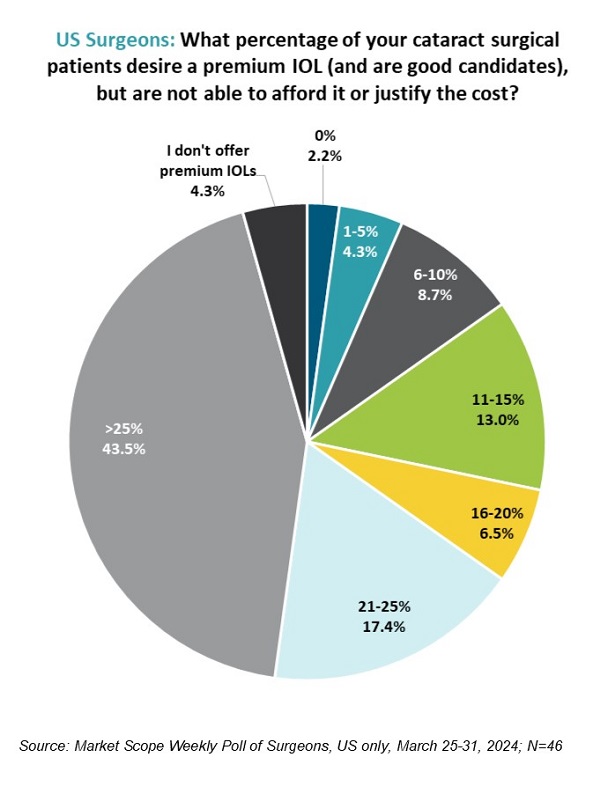
More News

ABVC BioPharma Sells Global Rights to Vitreous Substitute to ForSeeCon Eye for $30 Million Up Front
ABVC BioPharma announced March 26 that it had agreed to license its ophthalmology pipeline, including the vitreous substitute candidate Vitargus, to ForSeeCon Eye. ABVC, of Fremont, California, say...

US FDA Clears IND Amendment with New Mobility Test for Phase III Trial of Ocugen RP Gene Therapy Candidate
Ocugen announced April 8 that the US FDA had cleared the company’s investigational new drug (IND) amendment to initiate a Phase III clinical trial of OCU400, a modifier gene therapy candidate for r...
Want to Read Locked Articles?
Already Have an Account?
Register A Corporate Account
A corporate account gives you access to licensed reports and subscriptions, the latest news, a personalized dashboard, and weekly emails with news and data.